
The global weight-loss drug market has quickly become one of the hottest battlegrounds in healthcare. The explosive popularity of obesity drugs like Wegovy, Ozempic, and Zepbound has created one of the fastest-growing pharmaceutical markets in the world, dominated by giants like Novo Nordisk (NVO) and Eli Lilly (LLY). But now one smaller biotech company — Viking Therapeutics (VKTX) — is drawing attention as a possible surprise challenger, thanks to promising clinical data for its obesity drug candidate VK2735 and plans for potential commercialization.
Let’s find out whether Viking stands a chance in this heated market.
The Weight-Loss Drug Boom Comes With Growing Concerns
As someone with a fitness background, I personally remain deeply skeptical about relying on weight-loss drugs. Speaking from experience, I believe sustainable fat loss happens with nutrition, exercise, sleep and lifestyle changes rather than depending on drugs. And the concern is not just personal. While GLP-1 drugs have produced impressive results, many patients have reported serious side effects linked to drugs like Ozempic, Wegovy, and similar GLP-1 therapies. In fact, complications with these drugs have led to several lawsuits against Novo Nordisk and Eli Lilly.
Despite these concerns, though, one cannot ignore just how fast this market is growing. Experts indicate that the global weight-loss drug market could increase from $7.5 billion in 2025 to $67.2 billion by 2034. Now, Viking Therapeutics is emerging as one of the most closely watched names in the obesity-drug race.
Viking Is Betting Big on Weight-Loss Drugs
During Viking's first-quarter 2026 earnings call, management outlined an ambitious strategy for its obesity drug candidate VK2735, hinting that the weight-loss drug race could soon have a surprise challenger. Viking’s strategy appears to be very similar to Eli Lilly’s winning formula. VK2735 is a dual GLP-1 and GIP receptor agonist, which means it targets the same key pathways involved in hunger control, insulin regulation, and metabolism that helped make Eli Lilly’s Zepbound such a breakthrough success. The company said that the injectable version of VK2735 has shown “promising efficacy, safety, and tolerability across multiple clinical trials.”
In its Phase 2 VENTURE trial, patients showed up to 14.7% reduction in body weight after 13 weekly doses, with no signs of weight-loss plateauing during the study period. The most important factor to notice was the drug’s tolerability. Viking stated that the majority of side effects in the VENTURE study were considered as “mild or moderate” and resolved quickly. The drug is now undergoing a Phase 3 VANQUISH clinical trial which began in June of last year. Within that, two separate studies have begun, with VANQUISH-1 focused on adults with obesity while VANQUISH-2 targets adults with obesity and type 2 diabetes.
Viking aims to develop both injectable and oral obesity treatments, as it believes that many patients would choose to begin treatment with oral medication or switch to oral therapy following initial weight loss with injections. In the Phase 2 VENTURE study, the oral version of VK2735 resulted in considerable weight loss reaching up to 12.2% after 13 weeks. Viking also highlighted that 98% of drug-related “treatment emergent adverse events” were classified as mild or moderate. Viking is now planning to begin an oral Phase 3 trial in Q4 2026.
Besides VK2735, the company recently filed an IND for VK3019, its novel amylin receptor agonist program. The company believes this therapy could be a good addition to its broader obesity portfolio as early data shows amylin receptor activation may help regulate appetite and body weight. Viking intends to launch a Phase 1 clinical trial for VK3019 later this quarter, pending regulatory clearance.
To get ahead with potential commercialization, Viking has appointed Neil Aubuchon as its first Chief Commercial Officer. Aubuchon formerly served for nearly 17 years at Eli Lilly in leadership positions.
Viking’s aggressive expansion has led to increased R&D costs, which stood at $115.2 million in the first quarter. Consequently, net loss increased to $158.3 million in Q1, compared to $45.6 million in the year-ago quarter.
On the balance sheet, Viking ended the quarter with $603 million in cash, cash equivalents, and short-term investments. Management believes this should allow the company to complete its ongoing Phase 3 obesity trials while continuing development across additional programs.
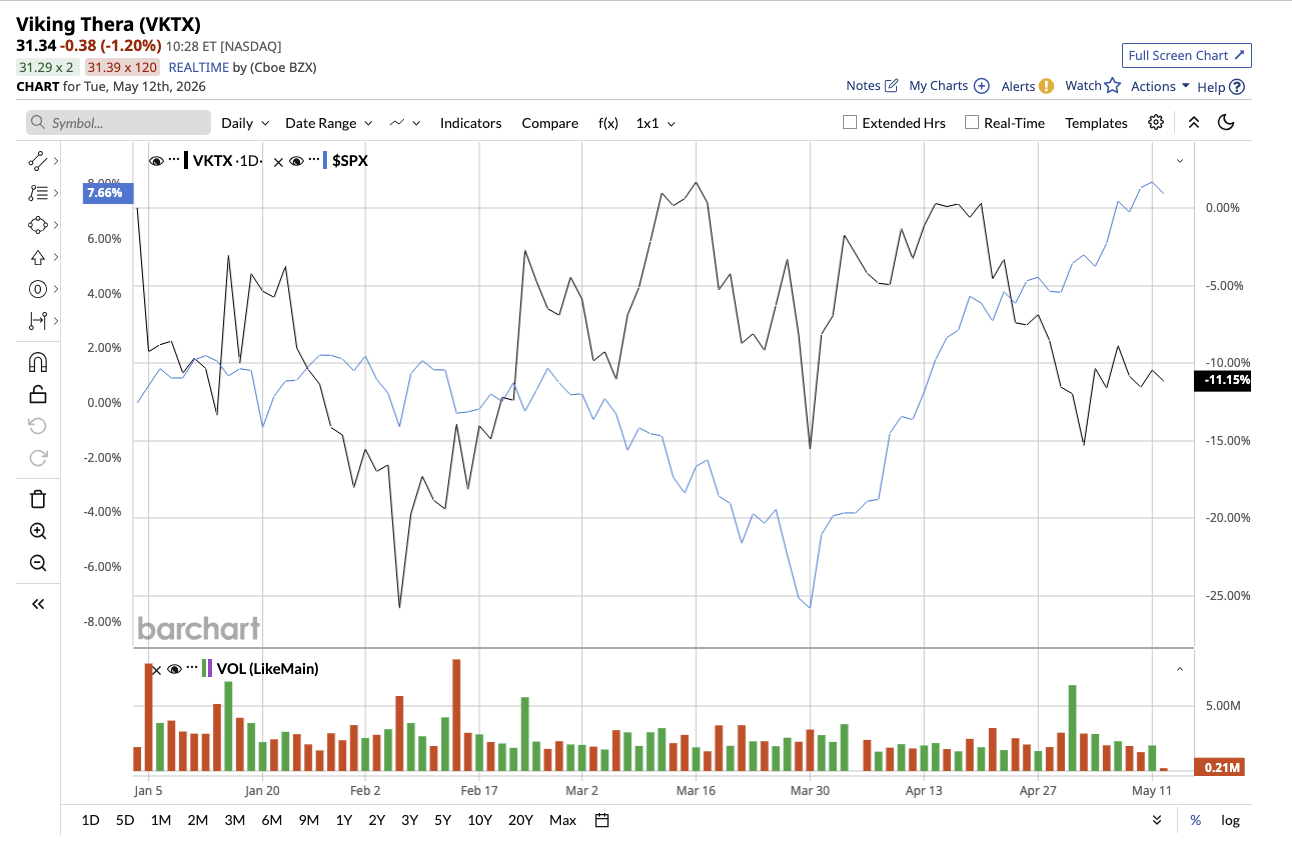
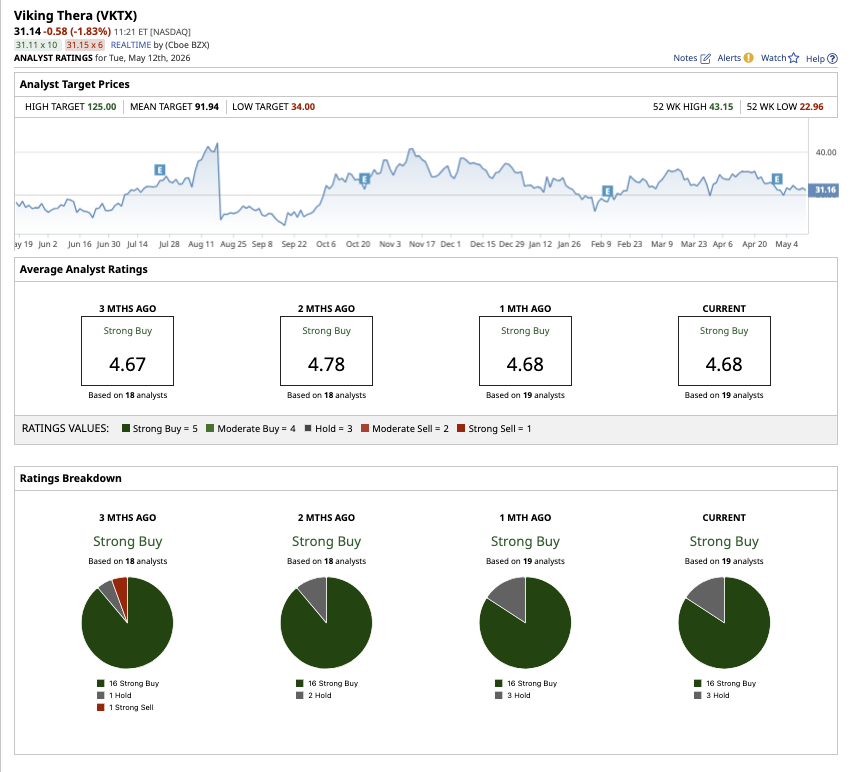
Despite the progress with its obesity candidate, Viking’s heavy losses are weighing on VKTX stock, which is down 10% year-to-date (YTD). Nonetheless, the average analyst target price of $91.94 suggests potential upside of 191% from current levels. Meanwhile, the high price estimate of $125 implies 296% potential upside over the next 12 months.
Is Viking Stock a Buy Now?
With a market capitalization of $3.68 billion, Viking Therapeutics remains a smaller player in a market dominated by the obesity-drug giants. For VK2735 to stand out, the company will need to prove that its treatment is different and more efficient over already successful drugs like Wegovy, Ozempic, and Zepbound.
If Viking has learned anything from the mounting side-effect concerns and legal scrutiny its rivals have faced, improving safety and tolerability could become one of VK2735’s biggest opportunities. The obesity market may ultimately be large enough for multiple winners. For long-term investors who can handle the volatility that comes with biotech stocks, Viking increasingly looks like a high-risk, high-reward growth story that may still be in its early innings.
On Wall Street, Viking stock has a consensus “Strong Buy" rating overall. Out of the 19 analysts that cover VKTX stock, 16 rate it as a “Strong Buy” while three analysts rate it as a “Hold.”
On the date of publication, Sushree Mohanty did not have (either directly or indirectly) positions in any of the securities mentioned in this article. All information and data in this article is solely for informational purposes. For more information please view the Barchart Disclosure Policy here.



/Broadcom%20Inc%20HQ%20photo-by%20Sundry%20Photogrpahy%20via%20iStock.jpg)
/AI%20(artificial%20intelligence)/3D%20Graphics%20Concept%20Big%20Data%20Center%20by%20Gorodenkoff%20via%20Shutterstock.jpg)
/An%20image%20of%20a%20Tesla%20humanoid%20robot%20in%20front%20of%20the%20company%20logo%20Around%20the%20World%20Photos%20via%20Shutterstock.jpg)