Shares of ProKidney Corp. (NASDAQ: PROK) soared 515% on Tuesday, triggering shockwaves across Wall Street after the company unveiled highly promising Phase 2 trial results for its experimental kidney disease therapy, Rilparencel. The one-day jump pushed the stock from under $1 to a close of $3.73, capping a wild session that saw it hit an intraday high of $4.86 on volume nearly 40 times its daily average.
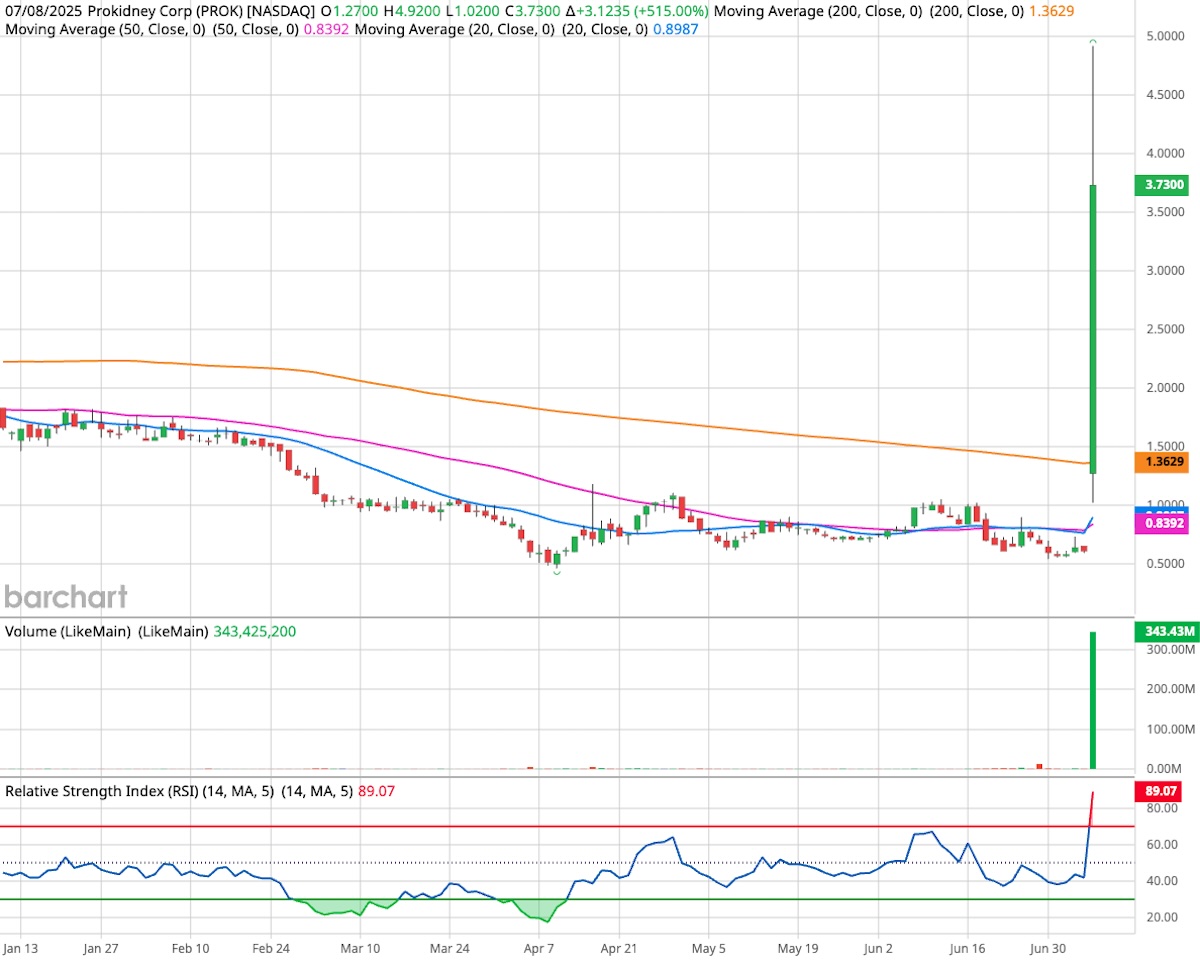
The surge follows the release of statistically and clinically significant topline data from ProKidney’s REGEN-007 study, which evaluated Rilparencel, a proprietary autologous cellular therapy, in patients with chronic kidney disease (CKD) and diabetes. Group 1 patients who received two scheduled injections showed a 78% improvement in their annual kidney function decline rate, a potentially groundbreaking outcome.
In the company’s official release, ProKidney CEO Bruce Culleton, M.D., stated:
We are very encouraged by the REGEN-007 topline results that demonstrated a robust improvement in eGFR slope following treatment with rilparencel in Group 1 as well as evidence of a dose response in Group 2. These data bolster our confidence in the design of our ongoing Phase 3 PROACT 1 study given the similarity between the dosing regimen in REGEN-007 Group 1 and PROACT 1.
A Breakthrough for Kidney Disease?
The eGFR (estimated glomerular filtration rate) slope, a measure of kidney function decline, improved significantly in Group 1, moving from -5.8 to -1.3 mL/min/1.73m² after Rilparencel treatment. This 4.6 point improvement translates to a 78% relative benefit and reached statistical significance (p<0.001). No serious adverse events linked to the therapy were reported, and the safety profile remained in line with expectations.
Group 2, which followed an alternate dosing schedule, showed a 50% improvement in eGFR slope, though the results did not reach statistical significance (p=0.085). Still, the findings suggest a potential dose-response relationship, a positive signal for future development.
ProKidney plans to submit full results to the American Society of Nephrology’s Kidney Week 2025 and has a Type B FDA meeting scheduled for this summer to confirm its regulatory strategy. If successful, the company could seek accelerated approval based on the eGFR slope surrogate endpoint.
| Annual eGFR Slope (mL/min/1.73m2) | ||||||||
| Group | N (my) | Pre inj | Post last inj | Absolute benefit | Relative benefit | |||
| 1 | 24 | -5.8 | -1.3 | 4.6 | 78% | |||
| 2 | 25 | -3.4 | -1.7 | 1.7 | 50% | |||
Analysts Are Divided
While retail investors and some biotech bulls embraced the news with enthusiasm, Wall Street’s reaction remains split.
Citi (NYSE: C) responded with optimism, raising its price target from $6 to $9 and reiterating its Buy rating. The bank said the early data “exceeded expectations” and upgraded the probability of Rilparencel’s success to 60%. Citi pointed to the clear efficacy in Group 1 and its close alignment with Phase 3 dosing protocols as reasons for increased confidence.
But Bank of America (NYSE: BAC) remains firmly on the other side of the fence. The firm reiterated its Underperform rating and $1 price target, warning that the stock’s 500%+ move “looks overdone.” Analysts cited the absence of a sham comparator, lack of a post-results investor call, and a potential gap between trial timelines and ProKidney’s capital runway as major concerns. They also raised questions about whether the addressable market would be large enough to justify a bullish stance without more compelling data.
Read Now: The Small Cap Gold Market is Breaking Out Aggressively — Here’s What to Do
High Hopes, But Real Risks
ProKidney’s RMAT designation (Regenerative Medicine Advanced Therapy) from the FDA gives it a clearer pathway to accelerated approval, but it doesn’t eliminate risk. The company is currently running its Phase 3 PROACT 1 trial, with a modified protocol focusing on Stage 4 and late Stage 3b CKD patients. The trial’s success is critical to the future of Rilparencel and ProKidney as a company.
Despite Tuesday’s dramatic rally, PROK remains well below its 52-week high. Its market cap, while sharply higher, still hovers around $175 million. The company does boast a solid cash position and no significant debt, but its ongoing cash burn rate poses a challenge if timelines stretch beyond expectations.
Meanwhile, competing names in the kidney disease space, such as FibroGen (NASDAQ: FGEN) and Vertex Pharmaceuticals (NASDAQ: VRTX), also saw gains, though more modest, up 4.6% and 1.5%, respectively. The excitement over ProKidney’s cell therapy approach adds to growing interest in regenerative medicine as a new frontier in treating chronic diseases.
Is PROK a Buy?
ProKidney’s recent results are undeniably exciting and could mark the start of a biotech turnaround story. However, investors need to proceed with caution. The company faces a high bar to success: navigating a complex regulatory environment, delivering repeatable results in Phase 3, and managing its financial runway along the way.
Until the FDA weighs in and full data becomes public, the stock will likely remain highly volatile.
For now, ProKidney offers a classic biotech dilemma: enormous upside potential if things go right, but equally large risks if they don’t. Investors chasing Tuesday’s momentum should stay nimble, and those with a long-term outlook should prepare for a bumpy ride.
As Dr. Culleton emphasized:
We also look forward to our upcoming FDA Type B meeting in the coming weeks to confirm our approach to eGFR slope as a surrogate endpoint for accelerated approval. This meeting represents an important step toward our goal of expediting rilparencel’s potential path to market in the U.S. where there remains a significant unmet clinical need in patients with advanced CKD and diabetes.
Bottom line: ProKidney’s Phase 2 data is a major win, but the path to approval and commercial viability remains steep. Investors should watch the FDA meeting closely and keep their eyes on Phase 3 progress before making a big bet.
Small Cap News Movers & Winner Deep Dive – By WealthyVC.com
We scan over 10,000 publicly listed stocks across all seven North American exchanges to uncover the market-moving news that actually matters—focusing on high-quality, liquid, growth-oriented companies in sectors attracting serious capital, like AI, blockchain, biotech, and consumer tech.
Each week, we publish Small Cap News Movers, a curated roundup of small and micro-cap stocks surging on meaningful catalysts. We break down what’s driving the move, tap into rumors swirling on social media, and surface sharp insights from both industry experts and retail sleuths.
From this list, we select one standout stock for our Small Cap Winner Deep Dive, released the next day, where we take a closer look at the fundamentals, narrative, and technicals that suggest this winner could keep running.
Powered by our proprietary 4-element, AI-driven analysis system, our goal is simple: cut through the noise, remove the emotion, and help investors dominate the small-cap market with momentum-driven strategies—completely free.
Sign up for email alerts to get the moves before our social media followers.
Read Next: Is This Tiny Biotech Stock Ready for a Massive Breakout?
Join the Discussion in the WVC Facebook Investor Group
Do you have a stock tip or news story suggestion? Please email us at: invest@wealthyvc.com.
Disclaimer: Wealthy VC does not hold a position in any of the stocks, ETFs or cryptocurrencies mentioned in this article.
The post ProKidney Corp. Skyrockets 515% on Trial Data, But is it a Buy? appeared first on Wealthy Venture Capitalist.




/A%20Palantir%20sign%20displayed%20on%20an%20office%20building%20by%20Poetra_RH%20via%20Shutterstock.jpg)

